Shake-responsive viscosity
What makes EOHILIA different
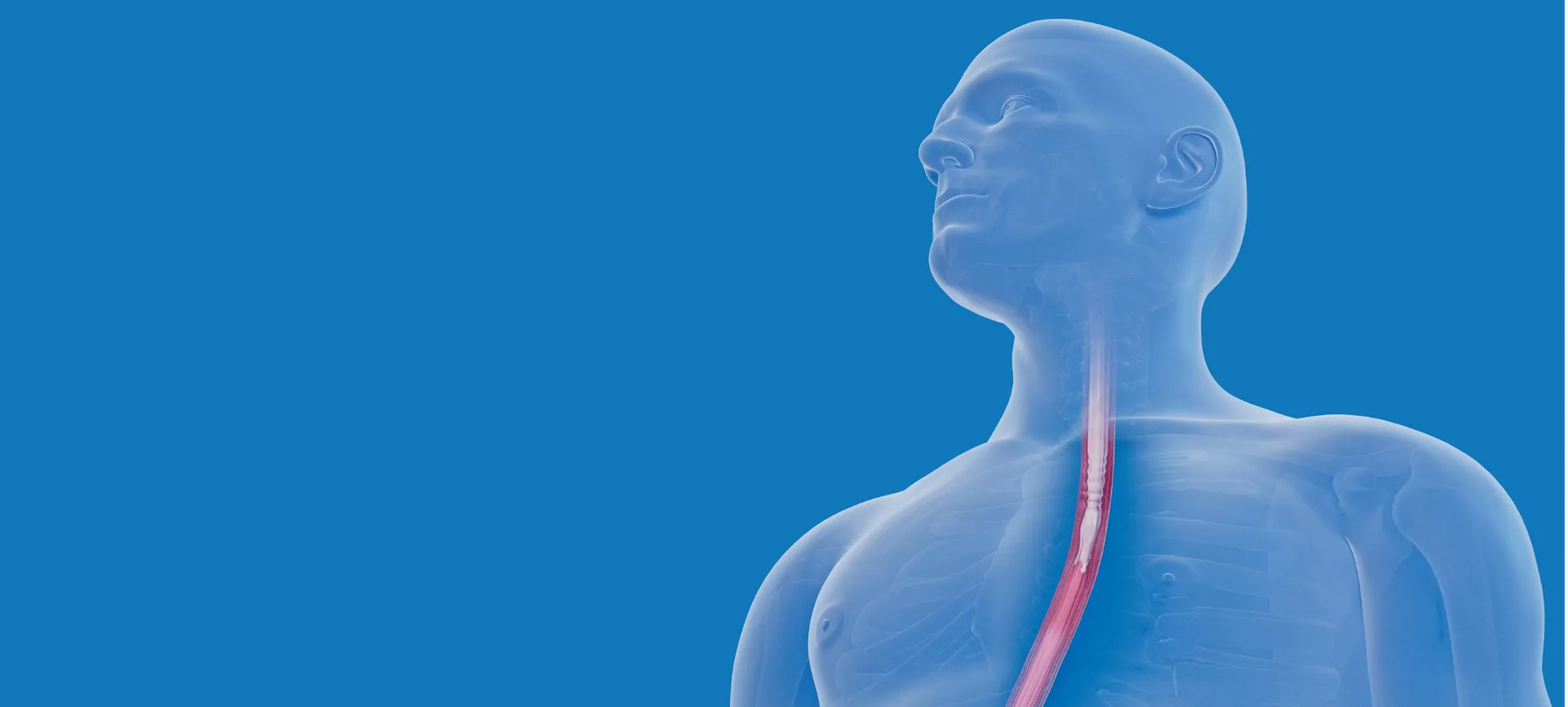
EOHILIA is designed with a viscosity that can change. At rest, EOHILIA is highly viscous. Upon shaking, it gets more fluid and then regains its viscosity to flow slower. This ability to go from high to low viscosity is known as thixotropy.1,2

The oral bioavailability in healthy subjects is ~14% under fasting state. Systemic effects may occur.
VIDEO TRANSCRIPT
While eosinophilic esophagitis (EoE) symptoms are unique in each patient, the underlying cause is esophageal inflammation, due to an increase in eosinophils in the esophagus.
That’s why it’s important to treat inflammation where it occurs.
EOHILIA (budesonide oral suspension) is a corticosteroid indicated for 12 weeks of treatment in patients 11 years and older with EoE. EOHILIA has not been shown to be safe and effective for longer than 12 weeks. EOHILIA is contraindicated for patients with hypersensitivity to budesonide. Serious hypersensitivity reactions, including anaphylaxis, have occurred with oral budesonide products. Please continue watching until the end of the video for full Important Safety Information.
EOHILIA is specifically designed for EoE.
It is the first and only oral 12-week, FDA-approved treatment.
Indicated for people with EoE 11 years and older.
EOHILIA is designed with a viscosity that can change. At rest, EOHILIA is highly viscous. Upon shaking, it gets more fluid; and then regains viscosity to flow slower.
The ability to go from low to high viscosity is known as thixotropy.
This physical property creates a novel budesonide formulation.
EOHILIA is designed to fit into patients’ pockets and their daily routines, with a twice-daily 12-week dosing regimen.
It is ready for on-the-go use in pre-prepared, single-dose stick packs with no mixing, measuring, or refrigeration required.
Remind your patients that EOHILIA should be taken as prescribed:
2 mg twice daily, once in the morning and once in the evening, for 12 weeks.
EOHILIA. Designed for EoE. Treat inflammation where it occurs.
Ingredients that matter
Anti-inflammatory effect
- Budesonide is an anti-inflammatory corticosteroid with a high glucocorticoid effect and a weak mineralocorticoid effect1
- Inflammation is a core component in the pathogenesis of EoE. Although the precise mechanism of corticosteroid actions on inflammation in EoE is not known, budesonide has a wide range of inhibitory activities against multiple cell types (eg, mast cells, eosinophils, neutrophils, macrophages, and lymphocytes) and mediators (eg, histamine, eicosanoids, leukocytes, and cytokines) involved in allergic inflammation1
- EOHILIA is 200 times more potent than cortisol and 15 times more potent than prednisolone1
Designed with patients in mind
- Excipients do not have any ingredients made from gluten-containing grain (wheat, barley, or rye)1
- Formulated with Avicel® RC-591, an excipient known for contributing high thixotropic behavior in pharmaceutical oral suspensions1,3
EOHILIA pharmacokinetics
Budesonide—the active ingredient in EOHILIA—has a high plasma clearance, 0.9 to 1.8 L/min approaching the estimated liver blood flow, suggesting that budesonide is a high hepatic clearance drug.1
- The mean plasma elimination half-life (t1/2) of budesonide after administration of EOHILIA was 3.3 hours1
- Following oral absorption, budesonide is subject to high first pass metabolism (80% to 90%)1
- The oral bioavailability of budesonide in healthy subjects is estimated to be 14% under fasting state1
References:
- EOHILIA (budesonide oral suspension) Prescribing Information. Takeda Pharmaceuticals, Inc.
- Lee CH, Moturi V, Lee Y. J Control Release. 2009;136(2):88-98.
- Pharma Excipients. AVICEL RC-591. Accessed August 16, 2024. https://www.pharmaexcipients.com/product/avicel-rc-591/.
VIDEO TRANSCRIPT
While eosinophilic esophagitis (EoE) symptoms are unique in each patient, the underlying cause is esophageal inflammation, due to an increase in eosinophils in the esophagus.
That’s why it’s important to treat inflammation where it occurs.
EOHILIA (budesonide oral suspension) is a corticosteroid indicated for 12 weeks of treatment in patients 11 years and older with EoE. EOHILIA has not been shown to be safe and effective for longer than 12 weeks. EOHILIA is contraindicated for patients with hypersensitivity to budesonide. Serious hypersensitivity reactions, including anaphylaxis, have occurred with oral budesonide products. Please continue watching until the end of the video for full Important Safety Information.
EOHILIA is specifically designed for EoE.
It is the first and only oral 12-week, FDA-approved treatment.
Indicated for people with EoE 11 years and older.
EOHILIA is designed with a viscosity that can change. At rest, EOHILIA is highly viscous. Upon shaking, it gets more fluid; and then regains viscosity to flow slower.
The ability to go from low to high viscosity is known as thixotropy.
This physical property creates a novel budesonide formulation.
EOHILIA is designed to fit into patients’ pockets and their daily routines, with a twice-daily 12-week dosing regimen.
It is ready for on-the-go use in pre-prepared, unit-dose packets with no mixing, measuring, or refrigeration required.
Remind your patients that EOHILIA should be taken as prescribed:
2 mg twice daily, once in the morning and once in the evening, for 12 weeks.
EOHILIA. Designed for EoE. Treat inflammation where it occurs.